HKUMed uses ancestral sequence reconstruction1 to reveal the evolutionary adaptations that enable avian influenza viruses to transmit in mammalian hosts
Researchers at the School of Public Health, LKS Faculty of Medicine of The University of Hong Kong (HKUMed), in collaboration with Duke-NUS Medical School (Duke-NUS) and St. Jude Children’s Research Hospital (SJCRH), retraced the natural avian-to-mammalian evolutionary process of the European avian-like H1N1 (EA) swine influenza viruses that jumped species in the late 1970s.
Researchers found that recombinant viruses representing different adaptive stages of the EA swine influenza viruses were reconstructed and characterised to identify step-wise molecular changes in the viral polymerase and nucleoprotein that facilitated efficient pig-to-pig transmission. The results suggest strategic coordination of surveillance and risk assessment activities may help to identify viruses with pandemic potential before they become fully adapted in the mammalian species. The findings are now published in the latest issue of Nature Microbiology, a world-leading scientific journal. [link to the publication]
Pandemic influenza viruses that possess sustained human-to-human transmissibility may emerge after interspecies transmission of animal influenza viruses. Understanding the evolutionary steps taken by avian influenza viruses as they adapt to mammals will help us to better monitor zoonotic influenza viruses with pandemic potential. A limited number of avian-origin influenza viruses have successfully established in mammalian hosts. These viruses offer the best opportunities to identify the molecular determinants for successful establishment of avian influenza viruses in mammals.
Research methods and findings
Led by Dr Yen Hui-ling, Associate Professor of the Division of Public Health Laboratory Sciences, School of Public Health, HKUMed; Dr Richard Webby, Department of Infectious Diseases of SJCRH; and Professor Gavin James Smith, Emerging Infectious Diseases Programme of Duke-NUS, the research team aimed to dissect and study viral evolution that had already occurred in nature by applying ancestral sequence reconstruction, based on sequence information of existing low pathogenic avian influenza virus and EA swine influenza viruses, to gain viruses representing different adaptive stages of the EA swine influenza virus as it transitioned from avian to swine hosts since 1979.
The research team demonstrated that efficient transmission of the EA swine influenza virus in piglets was gained through stepwise adaptations, with changes in viral attachment to sialyl receptors and increased replication potential in mammalian cells. Mutations in the viral polymerase (PB1-Q621R) and nucleoprotein (NP-R351K) found to be fixed in circulating EA swine influenza viruses after 1983 significantly increased viral transmissibility in piglets.
Research significance
The results suggest that ancestral sequence reconstruction is a suitable approach to investigate cross-species transmission and host adaptation of newly emerged pathogens. These results further suggest that there may be the opportunity to intervene at the early stage as avian influenza viruses adapt to mammalian hosts. Intervention strategies include viral surveillance at human-animal interface where spillover infections may occur, and risk assessment studies on newly emerged viruses before they become fully adapted to a new host. These strategies are most likely to be successful and represent the best use of limited preparedness resources.
About the research team
This multi-institutional collaborative study was led by Dr Yen Hui-ling, Associate Professor, Division of Public Health Laboratory Sciences, School of Public Health of HKUMed; Dr Richard Webby, Department of Infectious Diseases of SJCRH; and Professor Gavin James Smith, Programme Director (Interim) and Professor in the Emerging Infectious Diseases Programme of Duke-NUS. Dr Su Wen, Post-doctoral Fellow, Division of Public Health Laboratory Sciences, School of Public Health, HKUMed is the lead author of the study. Key local and overseas collaborators include Dr Michael Chan Chi-wai and Professor Malik Peiris, School of Public Health, HKUMed; Dr Robert G Webster, Department of Infectious Diseases, SJCRH; Dr Su Chuan-fang Yvonne, Emerging Infectious Diseases Programme, Duke-NUS; Dr Wu Chung-yi, Genomics Research Center, Academia Sinica, Taipei; Dr Stephan Pleschka, Institute of Medical Virology, Justus Liebig University Giessen, Germany; and Dr Kristien Van Reeth, Department of Virology, Ghent University, Belgium.
This study was supported by the US National Institute of Allergy and Infectious Diseases (NIAID) under Centers of Excellence for Influenza Research and Surveillance (CEIRS) contract no. HHSN272201400006C, the Theme-Based Research Scheme (Ref: T11-705/14N and T11-712/19-N) of the Research Grants Council under University Grants Council, Government of Hong Kong Special Administrative Region, and the Duke‐NUS Signature Research Programme from the Ministry of Health, Singapore.
[1] Ancestral sequence reconstruction is a powerful tool to infer primordial sequences from existing sequences.
About the School of Public Health, HKUMed
The School of Public Health, LKS Faculty of Medicine of The University of Hong Kong has a long and distinguished history in public health education and high impact research. With world leading research in infectious diseases as well as on non-communicable diseases of both local and global importance, the School has made significant contributions through its research and advocacy to improve the health of populations and individuals, both locally and globally. The School is a leading research and teaching hub in public health on influenza and other emerging viruses, control of non-communicable and infectious diseases, tobacco control, air pollution, psycho-oncology, behavioural sciences, exercise science, life-course epidemiology, population mental health, and health economics, health services planning and management. This work has informed international (e.g. the US Food and Drug Administration, Health Canada, the World Health Organization), national and local public health policies.
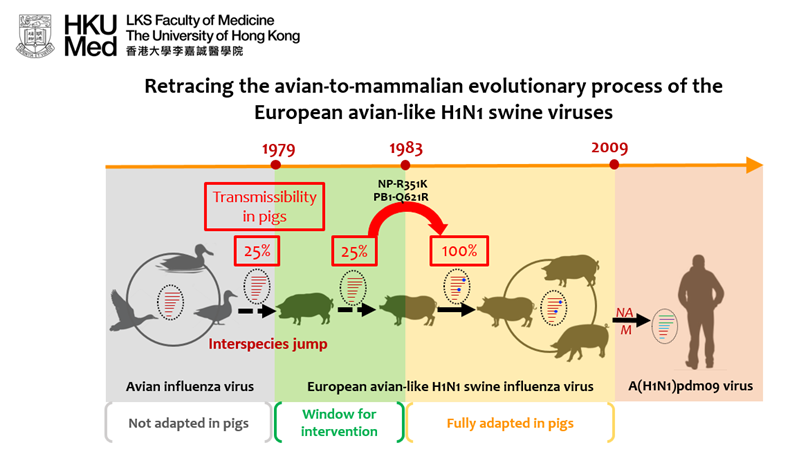
The European avian-like (EA) H1N1 swine influenza virus was derived from an avian influenza virus through interspecies jump that occurred prior to 1979. The EA swine influenza viruses have been established in pig herds in European and Asian countries since 1979. The EA swine viruses donated the neuraminidase (NA) and M gene segments to the A(H1N1)pdm09 virus that caused the 2009 influenza pandemic.
In this study, Su et al. investigated the molecular changes facilitated the avian-to-pig adaptation of the EA swine influenza viruses. Ancestral sequence reconstruction was used to gain viruses representing different adaptive stages of the EA swine influenza virus as it transitioned from avian to swine hosts since 1979. A key parameter for virus adaptation in a new host is its transmissibility.Transmissibility is assessed by counting the proportion of contact piglets (N=4) that become infected after co-housing with infected donor pigs in the same cage, a condition that allows all major modes of transmission to occur.
The research team found that the EA swine influenza viruses acquired amino acid changes in the viral polymerase (PB1-Q621R) and nucleoprotein (NP-R351K) that facilitated efficient pig-to-pig transmissibility after 1983. The results suggest a potential window for intervention (1979-1983) before the virus is fully adapted in pigs.







.png)
